Faculty Research: Tomer Avidor-Reiss
|
Research Areas: Cell Architecture and Dynamics; Microscopy and Imaging; Reproductive and Developmental Biology.
|
Professor Assistant Professor, Harvard Medical School, Boston, USA Phone: 419-530-1993 E-mail: Tomer.AvidorReiss@utoledo.edu Sites: http://loop.frontiersin.org/people/132963/overview https://www.researchgate.net/profile/Tomer_Avidor-Reiss http://sciencemission.com/index.php?page=tomer Publications |
Our research
We started our life as a single cell (the zygote) that was produced when the sperm fertilized the egg. This cell contained all the information to create an adult made of trillions of cells. Most of these adult cells must have two structures known as the centrioles, which are essential for building the cell’s antenna (the cilium) and skeleton (cytoskeleton) as well as for accurate cell division. How the zygote got its first two centrioles is a mystery and is the focus of our research.
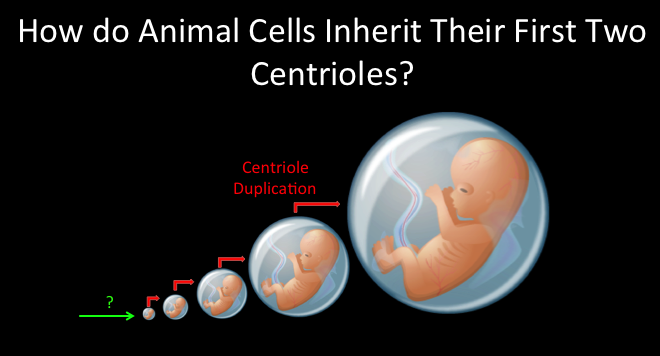
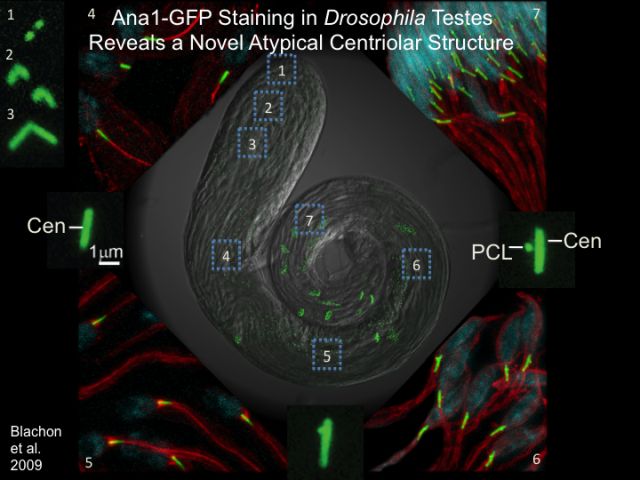
We know that the egg does not provide centrioles, so the origin of all the centrioles in our body is from the sperm of the father. However the sperm have only one centriole, so where does the second centriole come from? Using fruit flies as a model animal, we have discovered that the sperm has a new type of centriole that may solve this mystery and we named it the Proximal Centriole Like or PCL (see figure). This centriole structure is so small and different from the canonical centriole that it was missed in the past. We hypothesize that a similar atypical centriole structure is found in the human sperm and are currently searching for it. This is significant as, this atypical centriole may be the explanations to infertile couples that do knot know why they are infertile and have no treatment for their infertility.
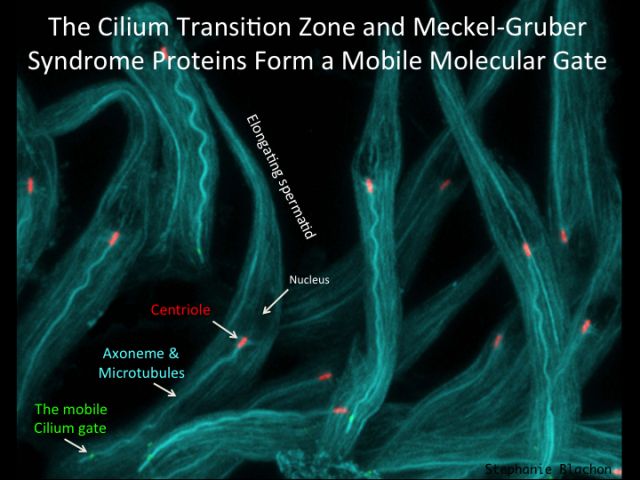
Ciliogenesis: During my postdoctoral research in the lab of Charles Zuker in the University of California, I studied the mechanism of cilia formation. To identify the genes required for cilia formation, I have developed a novel bioinformatic screen and found a large collection of genes required for cilia formation. To study the role of these genes in ciliogenesis, I have developed an experimental platform in Drosophila and showed, using genetic tools, that a subgroup of these genes encodes components of a ciliary transport pathway unique to cilia. This work has discovered many of the ciliary proteins that were studied in the last decade of ciliary research. One conclusion made in this study is that the flagellum of sperm cells are formed in a unique way that we named cytoplasmic ciliogenesis, as in this type of ciliogenesis the cilium axoneme is exposed to the cytoplasm. Recently we have discovered that cytoplasmic cilia are formed due to the migration of the cilium gate (the transition zone) away from the centriole exposing the axoneme to the cytoplasm. This finding opens a new challenge, to understand how the cilium gate, that is thought to be anchored to the cilium base, migrates along the cilium.
Centrosome assembly: My first work as an independent investigator was the discovery that Asterless/Cep152 is a key regulator in the initiation of centriole duplication. Before our publication, for nearly ten years, it was believed that Asterless was only essential for the function of the mature centrosome. Nonetheless, we showed that Asterless is instead essential for centriole duplication and that the absence of aster formation in asterless mutants is due to their lack of centrioles. Although initially our work encountered skepticism because it counters strong dogmatic belief, since then our conclusion has been verified by many groups and is now accepted as an undebated fact. My second line of investigation in centrosome assembly involves studying centrosome biogenesis in Drosophila using biochemistry. Many in our field considered it is too difficult and consequently, such biochemistry experiments have not been employed by others. After investing much time, thought, and effort, we successfully performed this type of study. By combining centrosome biochemistry with Drosophila genetics, we have found that Sas-4 forms several complexes of PCM proteins. We then showed that Tubulin, the building block of microtubules, functions as a switch like regulatory subunit that regulates the formation of the PCM complexes, and centrosome function.



