Xenon

Why is Xenon used in ion thrusters?
Contributor: University of Toledo Instrumentation Center
About the Display: As one of the noble gases, Xenon was chosen by the Instrumentation Center. The display box features a discharge tube in the shape of the element's symbol. The tube is filled with Xenon and powered by a transformer. When an electric charge excites the Xenon gas in the tube, the Xenon emits a blue or light lavender glow. The glow comes from an electron of the gas becoming excited from the energy. The excited electron leaves its electron shell and orbits the nucleus of the atom at a higher state. Eventually, the electron will return to its original state, releasing the extra energy as light.
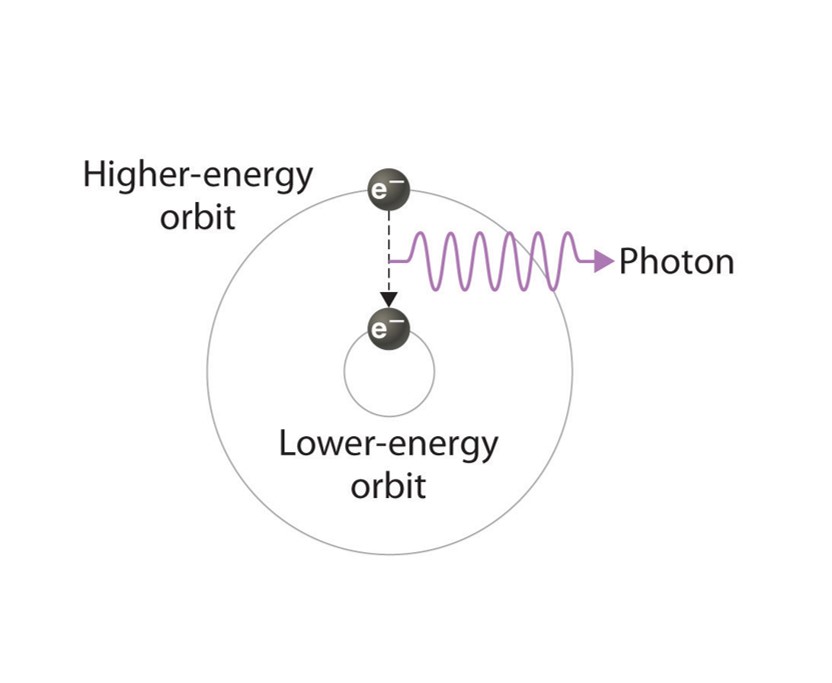
Above: A basic illustration of how an excited electron returns to its regular state and emits light. Picture courtesy of the open textbook library
Back to the Periodic Table
| <Previous element-| | |-Onward to the next element!> |
Symbol: Xe
Atomic Number: 54
Atomic Mass: 131.293 u
Electron Configuration: [Kr] 4d105s25p6
Year Discovered: 1898
Discovered By: Morris Travers, William Ramsay


