Faculty: Peter Andreana, Ph.D.

Department of Chemistry and Biochemistry
School of Green Chemistry and Engineering
Email: peter.andreana@utoledo.edu
Office: WO 2232B
Phone: (419) 530-1930
Fax: (419) 530-1930
Professional Background:
B.S. 1998: Brock University (Canada)
Ph.D. 2002: Wayne State University
NIH Postdoctoral Fellow: Harvard University 2002-2005
Publications
Group Web Site
Research Synopsis: Research interests in the Andreana group include natural product synthesis, carbohydrate-based
diversity oriented synthesis (DOS), carbohydrate-based vaccines and combinatorial
methods for high-throughput screening. The key underlying question for this research
program is: How do carbohydrates modulate cellular function? In order to address this
question, chemistry as the vector into biology is emphasized.
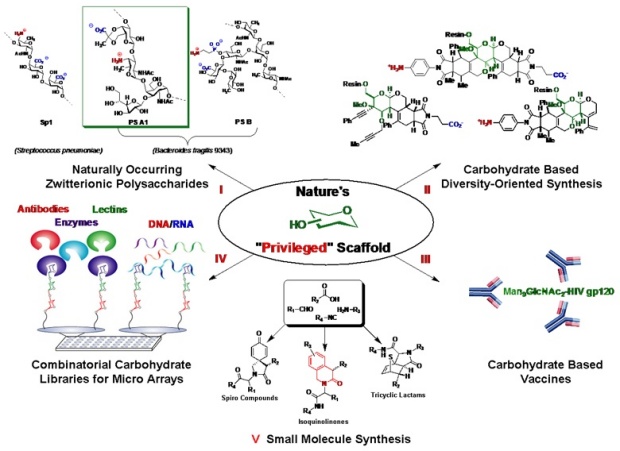
There are five major research directions our group currently pursues.
I. Synthesis of Naturally Occurring Carbohydrates / Polysaccharides
One objective is to prepare well-defined zwitterionic polysaccharides (ZPS), in which
the oligosaccharide is amenable to chemical modifications for the development of bioprobes.
The long term goal is to generate a clearer understanding of how these naturally synthesized
molecules illicit immune responses via the MHCII pathway (See figure pathway I).
II. Carbohydrate-Based, Diversity Oriented Synthesis
Another objective in our group seeks to examine a diversity oriented synthesis (DOS)
approach toward the construction of a class of compounds containing carbohydrate scaffolds
as potential therapeutic agents (See figure pathway II).
III. Carbohydrate-Based Vaccines
Current carbohydrate-based vaccines, while effective, do not protect against the carrier
protein and, therefore, an immunogenic response is heterogeneous in nature. Our objective
is to prepare well-known carbohydrate antigens, such as the well-defined polysaccharide
(Man9GlcNAc2-HIV gp120), in which the oligosaccharides are linked to a T-cell inducing
ZPS for the development of carbohydrate-based vaccines (See figure pathway III).
IV. Combinatorial Carbohydrate Libraries and Carbohydrate-Based Micro Arrays
To determine whether an alternating charge character is necessary to elicit a T-cell
response, the Andreana group will take advantage of the solid support, split-pool
concept of combinatorial chemistry and develop a carbohydrate-based library with electrostatic
charge character. It is of great interest to determine, systematically, how the electrostatic
charges of ZPSs interact with various modulators of APCs (See figure pathway IV).
V. Small Molecule Synthesis (Green Chemistry)
The proposed small molecule synthesis ties in extremely well with a platform for the
development of modulators (Chemical Genetics) of the immune pathway to determine mechanistic
insights of carbohydrate processing in the antigen presenting cell or dendrite cell.
The tyrosine kinase p56 (lck) is present in T-cells and is known to be required to
initiate the activation response from the T-cell receptor (TCR) intracellular domain
to other signaling proteins. T-cells that lack lck are unable to respond to stimulation
through the TCR (See figure pathway V).